[ad_1]
Credit score: Journal of Medical Investigation (2023). DOI: 10.1172/JCI166666
Researchers at Karolinska Institutet and Kanazawa College in Japan have made a brand new discovery that would enhance breast most cancers remedies. The researchers studied most cancers stem cells (CSCs), which might resist chemotherapy and kind new tumors. They discovered a kind of CSC that’s weak to medicine that concentrate on a pump within the cells. These medicine might doubtlessly kill these CSCs and assist sufferers with triple-negative breast most cancers (TNBC), a hard-to-treat type of breast most cancers.
The work is published within the Journal of Medical Investigation.
Breast most cancers is a posh and numerous illness that impacts thousands and thousands of ladies worldwide. One of many challenges in treating breast most cancers is that a few of the cancer cells have the power to outlive chemotherapy and kind new tumors. These cells are referred to as cancer stem cells (CSCs), and they don’t seem to be all the identical. Some CSCs have completely different options and behaviors than others, which makes them tougher to focus on.
“We used a way referred to as single-cell RNA sequencing to investigate the range of CSCs in a kind of breast most cancers referred to as triple-negative breast cancer,” says Affiliate Professor Theodoros Foukakis on the Division of Oncology-Pathology. TNBC is an aggressive and hard-to-treat type of breast cancer that doesn’t reply to hormone therapy or focused medicine.
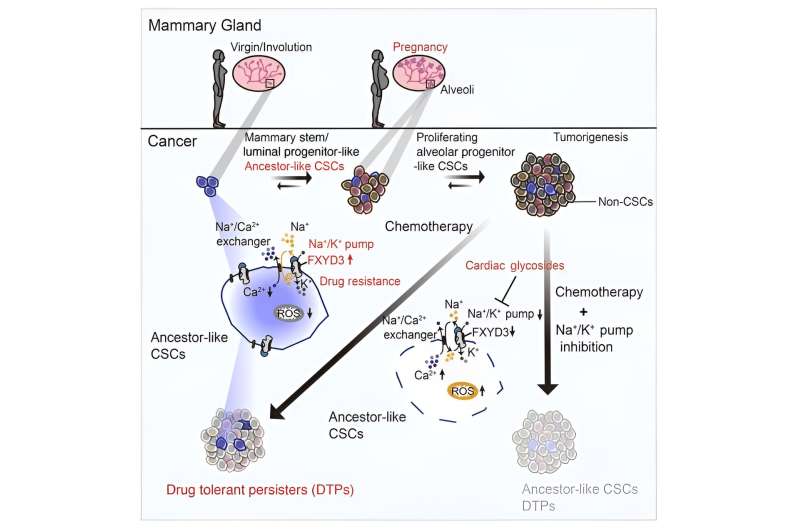
The researchers discovered that there’s a subpopulation of CSCs that has ancestral options, which means that they resemble the cells that kind the breast tissue throughout being pregnant. These CSCs are marked by a protein referred to as FXYD3, which is a part of a pump that regulates the steadiness of sodium and potassium ions within the cells. Curiously, following chemotherapy, these cells endured in resistant tumors however diminished in responding tumors.
The researchers found that FXYD3+ CSCs usually tend to evolve and proliferate, and that they’ve traits of alveolar progenitors, that are cells that kind the milk-producing glands within the breast. These cells are usually induced throughout being pregnant, however in TNBC they will contribute to tumor progress and unfold.
The researchers additionally discovered that FXYD3+ CSCs are extra proof against chemotherapy, and that they persist after therapy, making them potential sources of tumor recurrence. They recognized FXYD3+ CSCs as essential therapeutic targets, and instructed that eliminating them might enhance the prognosis of TNBC sufferers.
To check this concept, the researchers used medicine that inhibit the sodium–potassium pump, corresponding to cardiac glycosides, that are usually used to deal with coronary heart failure. They discovered that these medicine had been efficient in killing FXYD3+ CSCs, each within the laboratory and in animal fashions. In addition they discovered that these medicine might improve the impact of chemotherapy, and forestall the formation of latest tumors from FXYD3+ CSCs.
In abstract, the researchers have uncovered a paradoxical position of FXYD3+ CSCs in TNBC. These CSCs have ancestral options that make them extra adaptable and aggressive, but in addition extra weak to medicine that concentrate on the sodium–potassium pump. By exploiting this weak point, the researchers have proposed a brand new technique to get rid of CSCs which can be drivers of tumor plasticity and chemoresistance in TNBC. This might result in higher outcomes for sufferers with this difficult illness.
Extra info:
Mengjiao Li et al, FXYD3 functionally demarcates an ancestral breast most cancers stem cell subpopulation with options of drug-tolerant persisters, Journal of Medical Investigation (2023). DOI: 10.1172/JCI166666
Supplied by
Karolinska Institutet
Quotation:
New strategy to kill breast most cancers stem cells which have ancestral options and resist chemotherapy (2023, November 24)
retrieved 25 November 2023
from https://medicalxpress.com/information/2023-11-breast-cancer-stem-cells-ancestral.html
This doc is topic to copyright. Aside from any honest dealing for the aim of personal research or analysis, no
half could also be reproduced with out the written permission. The content material is offered for info functions solely.
[ad_2]
Source link




Discussion about this post