[ad_1]
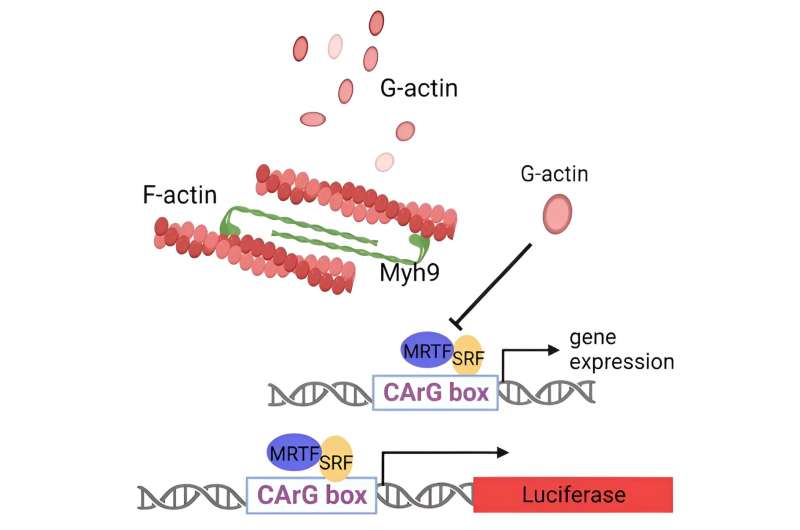
Higher panel: Diagram illustrating the interplay between Myh9, Actin and MRTF-SRF sign pathway. Myh9 (Myosin IIA) fashioned bipolar filaments binding to F-actin which may depolymerize into monomer G-actin. G-actin can bind to SRF cofactor MRTF to inhibit MRTF-SRF transcriptional exercise. Decrease panel: the diagram of SRF-RE Luciferase reporter to look at the exercise of MRTF-SRF pathway. Credit score: Nature Communications (2024). DOI: 10.1038/s41467-024-44778-2
A new study has discovered that the circadian clock—which synchronizes physiological and mobile actions with the day-night cycle and is mostly regarded as tumor suppressive—in reality has a contextually variable function in most cancers.
“A variety of proof means that the organic clock is damaged in most cancers cells, so we anticipated its disruption would gas tumor growth in mouse fashions of melanoma,” mentioned Chi Van Dang, scientific director of the Ludwig Institute for Most cancers Analysis, who led the examine with Analysis Affiliate Xue Zhang. “However, opposite to our expectations, we discovered that lack of a gene referred to as Bmal1—a grasp regulator of the mobile clock—as a substitute dampens the expansion of melanoma tumors in mice.”
The researchers’ exploration of why that is the case, described within the present concern of Nature Communicationshas uncovered beforehand unknown mechanisms of tumor suppression and resistance to immunotherapy in melanoma.
“Our findings might have implications for the design of chronotherapies, which search to coordinate most cancers remedies with organic rhythms to enhance their efficacy and mitigate their unwanted effects,” mentioned Zhang.
The circadian clock synchronizes the physiology of the physique and the metabolism of every of its individual cells with the photo voltaic cycle, alternating phases of vibrant exercise with intervals of relaxation and recuperation. In healthy cells the depth of metabolic exercise oscillates with the organic clock. Most cancers cells, nonetheless, rewire their metabolism to make sure a gentle provide of the uncooked supplies required for unrestrained proliferation. For the reason that intervals of relaxation enforced by the circadian rhythms can undermine that agenda, in addition they have a tendency to interrupt their organic clocks.
The Dang lab, which has made seminal contributions to our understanding of most cancers cell metabolism, has additionally explored hyperlinks between the metabolic rewiring of most cancers cells and their dysfunctional clocks. Most notably, research led by Dang have proven that oncogenic variations of the Myc gene, a grasp regulator of most cancers metabolism, suppress Bmal1 to disable the cell’s metabolic brakes and gas proliferation.
The sluggish development of melanoma tumors missing Bmal1 was not per that discovering. On the lookout for an evidence, the researchers examined how gene expression modified in these cells and noticed that the lack of Bmal1 compromised the exercise of one other grasp regulator of gene expression, HIF1, that helps most cancers cells adapt to low-oxygen situations prevalent in lots of strong tumors. Additionally they confirmed that expressing HIF1 at excessive ranges in melanoma cells that lack Bmal1 restores the flexibility of these cells to generate tumors.
However would Bmal1 be tumor suppressive if expressed at sufficiently excessive ranges? Apparently not, and it seems that this has nothing to do with Bmal1’s main function as a regulator of gene expression. A model of the protein that might do the whole lot abnormal Bmal1 does besides bind to DNA—which it should do to control circadian gene expression—was an excellent higher driver of tumor development.
“That discovering was a little bit of a head-scratcher,” mentioned Dang. “It did not match with something that we might count on.”
To resolve the puzzle, the researchers carried out a sweeping evaluation of protein interactions in Bmal1-overexpressing cells. They discovered that Bmal1 binds and sequesters a protein concerned in cell motility, MYH9, that’s regarded as a tumor suppressor—although the way it exerts that impact was, till now, unclear. Other than figuring out the mechanism by which MYH9 suppresses tumorigenesis, the researchers present that deleting the MYH9 gene in melanoma cells boosts their means to type tumors in mice.
Bmal1’s sequestration of MYH9 induces a number of adjustments within the molecular signaling networks of cells. Its internet impact, nonetheless, is to revert melanoma cells right into a developmentally immature “mesenchymal” state that’s seen in human tumors and is related to resistance to immunotherapy. Certainly, the researchers present that melanoma cells expressing excessive ranges Bmal1 type immunotherapy-resistant tumors within the mice.
With the assist of Ludwig Harvard’s Sandro Santagata and Peter Sorger, who lead the Ludwig Tumor Atlas mission, the researchers employed superior microscopy to indicate that tumors over-expressing Bmal1 bear molecular markers of the mesenchymal state. Additionally they demonstrated that these tumors in mice resist immunotherapy as a result of they’re insufficiently infiltrated with immune cells that focus on cancer cells and have a tendency to accommodate immunosuppressive cells as a substitute.
“Our findings general present that Bmal1 drives tumorigenesis and therapeutic resistance in melanomasuggesting that the impact of this regulatory protein on tumorigenesis almost definitely varies between most cancers varieties,” mentioned Dang. “They could additionally assist clarify a current discovery that prostate tumors expressing Bmal1 at excessive ranges resist androgen remedy—although that should be confirmed with additional analysis.”
Past that, the findings have opened new avenues for analysis on mechanisms of tumor plasticity and cancer drug resistance that the Dang lab will now pursue.
Extra data:
Xue Zhang et al, Cell state dependent results of Bmal1 on melanoma immunity and tumorigenicity, Nature Communications (2024). DOI: 10.1038/s41467-024-44778-2
Offered by
Ludwig Cancer Research
Quotation:
How a timekeeping gene impacts tumor development relies upon solely on context (2024, January 24)
retrieved 24 January 2024
from https://medicalxpress.com/information/2024-01-timekeeping-gene-affects-tumor-growth.html
This doc is topic to copyright. Aside from any honest dealing for the aim of personal examine or analysis, no
half could also be reproduced with out the written permission. The content material is offered for data functions solely.
[ad_2]
Source link




Discussion about this post